We all take the ease of peeling open and resealing packs of double-stuff Oreos for granted. The plastic packaging that maintains the freshness of our favorite snacks and foods has become so ubiquitous it doesn’t even register as existing until we try to open that hotdog pack with our bare hands or reseal that Oreo package (and fail to do so). But the manufacturers of these sorts of films and plastic products face issues that can’t be solved by grabbing a pair of scissors, passing the hotdogs to your roommate who has less greasy fingers than you, or using duct tape to keep those cookies fresh.
Issues of laminating, sealing, and printing on films and thin, extruded plastics like polypropylene and polyethylene are extremely common for manufacturers. Treatments of the film surfaces drive the primary action to prevent delamination, ink not sticking, and leaky seals/stale cookies. These treatments are effective at helping promote adhesion between plastics and adhesives to make sure they stick together. But, without proper controls, it’s difficult to know when the treatments are trending towards ineffectiveness or are being hampered by unforeseen hurdles (i.e. contaminants on the surface that prevent good adhesion but their origin hasn’t been traced).
The packaging made with these plastic films is extremely valuable as they can keep food fresh by blocking UV rays that can cause spoilage, keep moisture away from paper products, and create a barrier against bacteria, and they come with a whole lot of other safety mechanisms that allow us to ship and preserve so many items we use and rely on every day.
Rethink your adhesion manufacturing processes with Surface Intelligence.
A common and sometimes overlooked threat to this plastic sealing and laminating properly is an internal issue. There are very necessary additives combined with the raw polymeric materials that serve various functions but can cause problems under certain conditions and make adhesion inconsistent and unpredictable.
It is possible to get these additives under control and spot them before they can disrupt a plastic film production process. Doing so requires understanding how these additives make their way to the surface of plastics (when they are typically buried in bulk material, several molecular layers deep in the plastics) and using the right technique to identify them when they do show up where they don’t belong.
What Additives are Used in Plastic Films and Why?
Polymers have innate properties that make them extremely useful for many applications. But some properties may need to be enhanced in some way. This is where additives come in.
Get more information on how to get to the bottom of issues related to controlling surface treatment for bonding, printing, or sealing polymer films. Download our eBook: Checklist: Adhesion Failure Root-Cause Analysis for Manufacturers.
Here’s a list of common additives used to make plastics do all the things we rely on plastics to do on a daily basis:
- Plasticizers
- Lubricants and mold release agents
- Scratch and mar additives
- Antistats
- Compatibilizers
- Impact modifiers
- Reinforcing fillers; coupling agents
- Pigments
- Blowing agents
- Antioxidants/stabilizers
Some of these are used as aids in the manufacturing process - making sure the plastics don’t stick to molds and the like. Some are for aesthetic purposes and help maintain the appearance of the plastic - like helping it keep its intended shape, keeping oxidation from degrading the material, and preventing unsightly scratches or blemishes. Others in the list are used to reduce static on the surface, help foams expand to a prescribed firmness (blowing agents), and make sure the polymer blends that might be used are able to work together in the plastic that is derived from their amalgamation.
There are many more additives used in polymers that we won’t get into here, but here’s a more exhaustive list that also outlines their functions.
Most of these additives are what are called non-migratory. The non-migratory additives are not generally a problem because they typically disperse throughout the polymer very stably and don’t interfere with the surface in a way that causes disruptions to adhesion.
But lubricants, like slip and mold release agents, scratch and mar additives, and antistats, are designed to be migratory. They need to be present on the surface of the plastic, or else they aren’t doing their job. There are issues that can crop up with these, but for the purposes of this blog post, we’re going to focus on the one additive in the list I haven’t mentioned yet: plasticizers.
Plasticizers function to reduce the stiffness or the Tg (glass transition temperature) of a polymer. Their function depends on being dispersed uniformly throughout the bulk of the polymer. They need to show excellent solubility in a polymer, but if they migrate to the surface, that's a problem when it comes time to adhere to that surface.
Blooming of Polymeric Additives
All polymers are made up of molecule chains that take different net-like, cross-linked structures based on their type.

Common plastic films like polyethylene and polypropylene are thermoplastics. So chains that make up these polymers have a loosely piled structure, somewhat resembling a plate of spaghetti. This structure gives them a high molecular mobility, meaning that substances within the polymer can move around and diffuse from the bulk of the polymer to the surface and vice versa; substances can creep into the bulk of the plastic from the surface.
Blooming is the term given to the movement of substances in the bulk material to the surface.
When a plastic material is fabricated into an object like a film or any other form, e.g. via molding, extrusion, or thermoforming, the surface is not in equilibrium. The process creates a surface that didn’t exist before, and this means that the fresh surface has a low concentration of substances in the bulk material, like plasticizers. These surfaces also have a very high surface energy. A high-energy surface is a surface that is very reactive and very interested in bonding with molecules it may come into contact with. The higher the energy a surface has, the quicker it wants to reach equilibrium and stop being so reactive.
The rate and extent to which the polymer system moves toward equilibrium depends on:
- The concentration of additive in polymer - High concentration favors blooming
- Mobility of additive: molecular size; the free volume of polymer - Small additive molecules have higher mobility
- Solubility of the additive: molecular structure (solubility parameter)
- Temperature: affects mobility and solubility - High temperature increases mobility more than the solubility
- The surface energy of material - High surface energy materials favor blooming
Blooming affects important surface properties that are critical to anything you want to do to the surface. If you want to bond to it, paint it, bring it in contact with the human body, bring it in contact with foodstuffs, create a reliable seal, and print on it, then you need to pay attention to what additives are blooming to your surface during production.
What You Need to Know About Corona Treatment of Polymers
Even though a polymer with a high surface energy can be susceptible to blooming, to make a surface that is reactive enough to adhere to another surface, the surface energy needs to be raised. The molecular chains themselves that make up these polymers (as seen in the image earlier) are quite stable and require treatment to break them up in order to create a reactive surface. When the chains are broken, they want to regain equilibrium, and they attract substances to themselves, which makes them highly amenable to adhesion after treatment.
The treatment method of choice for polymer films is corona treatment. The treatment works by discharging high-voltage, high-frequency electricity from an electrode in a ceramic tube that runs the length of the roll of film being treated. The electricity is sent through the film to an electrically-grounded, metal roll called the treater roll, that the film is wrapped around. This interaction between the electrode and the metal roll creates a visible flash on the surface of the film as it moves between the two components. The results, however, are completely invisible to the human eye.
What is occurring is called surface activation. The electricity breaks the molecular chains on the surface of the film and creates a surface with high energy. The innate stability of polymers means they have intrinsically low surface energy. The chains may have a relatively loose structure, but the chains themselves are not easily severed.
The corona treatment is not a solution in and of itself without a verification step that measures the surface energy. Making sure the surface of the film is at the most optimum energy level for adhesion when it comes time to bond, print, or laminate is crucial to know you are creating a reliable bond.
One of the ways surface energy is typically tested is by using dyne inks. Dyne inks have a plethora of drawbacks to them which you can read about here. For polymer films, one of the biggest reasons not to use dyne inks is their lack of sensitivity. When additives bloom to the surface, they may only move the needle a small amount. This is a significant amount, but dyne inks won’t be able to tell the difference. They often can’t even tell the difference between a treated surface and a non-treated one.
Contact angle measurements are another solution that gives you a number correlated directly to the molecular characteristics of the film surface. This technique works by measuring the amount to which a tiny drop of water (usually) spreads out on the surface or constricts into itself, creating a taller droplet and, therefore a higher contact angle. A high contact angle indicates low surface energy. The water is sensitive to the changes in the attractive forces all objects have with each other that are altered through corona treatment. Water is also sensitive to substances blooming to the surface to establish equilibrium and lower the surface energy.
Optimize the power of next-gen connectivity with data & surface intelligence.
Another aspect of treatment that needs to be closely watched is the amount of time lapsing between the application of the corona treatment and the application of the adhesive or bonding step.
The chart below shows the relationship between treatment and time and how quickly the surface of flame-treated polypropylene (which is a process similar to corona treatment) can go back to the energy level it was at prior to treatment.
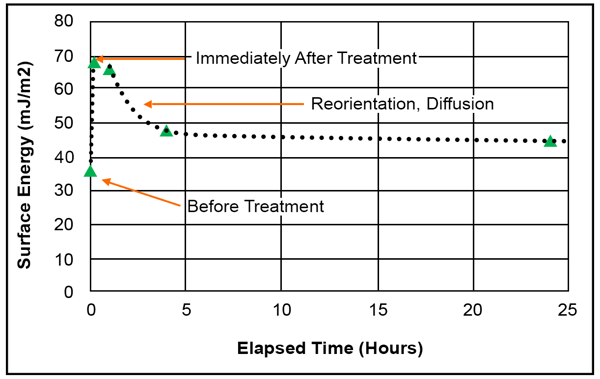
One reason for this degradation is due to the concept of molecular mobility, which was briefly mentioned earlier. This quality of polymers allows substances to seep into the bulk material from the surface. When a surface is treated, it is no longer in equilibrium, and the highly reactive molecules on the new, reactive surface will want to work their way down into the polymer and even out the concentration of molecules.
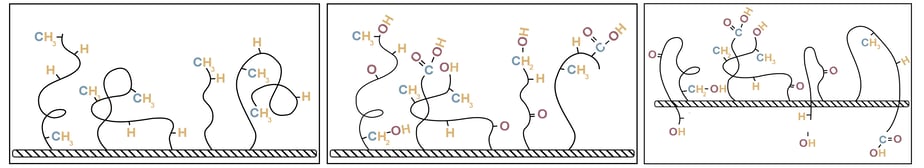
The three images below show a polymer surface before treatment, immediately after treatment, and then after sitting around for a few hours. The untreated surface just has normal hydrocarbon chains on it. The treated surface then has reactive sites through the breaking of the molecular chains and attaching oxygen atoms. These reactive groups are mobile and will then make their way into the bulk of the polymer, leaving the surface less active and less able to form strong bonds with another substance.
Often, the companies that are producing the final plastic products are not the same companies formulating the polymers or even molding them into shape. It’s extremely important to have an objective, quantitative and verifiable specification on polymer surface quality for all incoming materials. Knowing the reactivity of a polymer surface at each stage of production is a powerful quality control measure that can provide a greater level of predictability in your adhesion process.
In order to halt the deleterious effects of additive blooming, implementing a holistic approach to surface quality management is your best defense. Being able to recognize when blooming is occurring will allow you to bond, print, laminate, and seal with the utmost confidence.
It may be necessary to have a Materials Science expert that is familiar with your manufacturing process to give you insight into whether blooming is occurring and what contact angle measurements indicate the presence of additives on the surface. This level of granular understanding of your own process can help optimize the entire operation. It’s like the old saying goes, “you can’t manage what you don’t measure.”
For a deeper dive into blooming and its effect on adhesion processes, watch this webinar on the subject. For more information on how to get to the bottom of issues related to surface treatment, bonding, printing, sealing, or any of the processes discussed in this article, download our eBook: Checklist: Adhesion Failure Root-Cause Analysis for Manufacturers.





