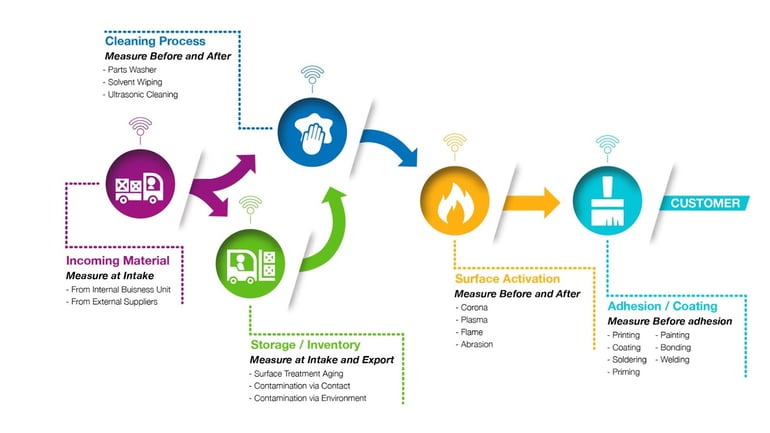
Today, manufacturing requires a paradigm shift from reactionary whack-a-mole problem correction to taking a holistic approach. The mutually reliant elements of manufacturing are interwoven in both obvious and invisible ways. From supply chain communication to equipment dependability and from process design validation to collecting accurate predictive performance data, manufacturing processes need smart sensors to gather useful analytics from beginning to end.
Adhesion, cleaning, and assembly processes begin long before parts enter the production facility and continue long after the final products make it into the hands of the end users.
Businesses in any industry must implement success metrics that are sensitive to the decreasing tolerance for contamination and low-quality parts in order to ensure continuity and achieve success. This approach will lead to a thriving yield that everyone desires. It is crucial for companies to be objective when defining these metrics in order to achieve their desired results.
Manufacturers are rightfully concerned about controlling three fundamental and overarching business dimensions: lowering costs, maximizing value, and minimizing risk.
These are not mutually exclusive, and focusing on one will surely have an impact on the others.
Rethink your adhesion manufacturing processes with Surface Intelligence.
Some of the factors that contribute to how well manufacturers balance cost, value, and risk management are waste (in the form of rework, downtime, and scrap), efficiency (keeping the rate of production at sustainable but ambitious levels), and productivity (ensuring yield is high and of high quality). Some setbacks, like recalls, can have a simultaneously detrimental effect on all three.
In order to help manufacturers across the industrial landscape rewire their thinking about production processes, we’ve come up with lists of Critical Control Points (CCPs) that are typically the sources of issues that hold manufacturers back from achieving their goals. These CCPs are interrelated steps that, when properly monitored, reveal the points at which quality decreases, leading to failures downstream.
Manufacturers have robust quality management systems, and yet they still experience loss in the form of waste, inefficiencies, and lagging productivity.
Honing in on surface cleaning, treatment, and preparation processes offers insight into often unquantified process steps that have a tremendous impact on the reliability of the end product.
Optimizing cleaning, treatment, and preparation processes has the knock-on effect of creating the positive outcomes manufacturers are looking for.
The following lists look at the automotive, aerospace, and consumer goods industries to show where manufacturers might not be looking to increase their margins in value expansion, risk mitigation, and cost reduction. Next week, we’ll take a look at the medical device, electronics, renewable energy, and flexible packaging industries.
Automotive Manufacturers Need a Reliable Way to Quantify the Cleanliness of Their Surfaces
Automotive assembly processes are becoming more and more sophisticated and less reliant on traditional joining techniques and technologies. As mechanical fasteners give way to adhesive bonding, knowing for certain that surfaces are clean enough to support strong adhesion is vital to success.
For more information on what steps to take to actually get to the root cause of adhesion and cleanliness issues, download our free eBook: Checklist: Adhesion Failure Root-Cause Analysis for Manufacturers
Here are a few process controls that need to be in place to guarantee the entire process is producing the most value while also reducing the costs inherent in rework, warranty claims, and reputation-damaging failures:
- Verify readiness of high-performance specifications composite surfaces for bonding
- Validate anti-fog coating qualities on headlights
- Detect contaminants (such as mold releases) on machined parts
- Verify the cleanliness of sealing surfaces in aluminum castings prior to bonding
- Verify a polymer’s readiness to be painted or bonded by validating treatment techniques such as flame and plasma treatments
- Ensure LIDAR sensor longevity through sealing validation and hydrophobic coating testing
- Validate high cleanliness levels required for FIPG (formed-in-place gaskets)
- Guarantee proper finishing of automotive interiors
- Validate clean surfaces on the engine block for RTV sealing
- Monitor and optimize the wash process for machined parts to ensure cleaning fluids are changed at the proper intervals to maintain the integrity of the parts being washed
- Measure brake and rotors for contamination to ensure coatings properly adhere
- Verify appropriate flame treatment of TPO used in bumper fascias, door panels, etc.
- Monitor bonding strength of composite materials
- Measure surface preparation prior to coating glass
RTV Sealing Application Example
As an example of how measuring cleanliness affects the performance of the finished product, let’s consider the fact that RTV sealing is increasingly replacing traditional gaskets. With this change, a reexamination of surface preparation processes must follow: this includes knowing and monitoring the cleanliness of the surface to ensure a successful bond. Moreover, RTV silicone requires a high-energy or very clean surface in order to form a strong bond.
So, when an automotive company was experiencing engine leaks in the field, they were forced to evaluate their sealing processes. Brighton Science partnered with them, and after evaluating their process, it was revealed that there was a huge variability in surface cleanliness measurements across dozens of parts. The variability in these measurements showed ranges of low surface energy resulting from a number of different contamination elements: heavy residual oils on the surface left over from the die casting process; washing systems designed to remove machining chips were not removing the detrimental residual contaminants; and, lastly, rust preventative—as a low energy coating—being used did not interact appropriately with RTV silicone.
With the new process of RTV sealing came the necessity to change several other processes. An appropriate surface treatment and washing process was needed to increase surface energy by properly removing molecular-level contaminants. Rust preventatives must be higher energy coatings to not interfere with the RTV bonding. Furthermore, the manufacturers implemented a quantitative cleanliness inspection system on the factory floor as an in-place method of monitoring surface cleanliness levels.
Aerospace Manufacturers Need Their Processes to be Suitable for Advanced Materials
The aerospace industry knows it must meet high specs to create an aircraft that is safe, reliable, and resilient. As aluminum and titanium materials are more often replaced with stronger, lightweight composites, which require adhesive bonds, knowing that bond will stick is pivotal. The strength and success of that adhesive bond depend on properly preparing the composite surface.
Here are critical processes that need to be protected by objective control measures that guarantee outcomes through predictive analytical evaluation:
- Verification of surface cleanliness for composite adhesive bonding and repair
- Verification of successful adhesion of aircraft mechanical fasteners that utilize adhesive bonding
- Verification of bonded repairs of wings, fuselages, and helicopter rotor blades
- Verification of metal cleanliness prior to brazing, welding, and heat treating
- Development of appropriate surface preparation processes
- Development of parameters for surface treatments
- Development of surface preparation techniques to train maintenance and repair technicians
- Monitoring of surface preparation processes such as solvent wiping, grit-blasting, pneumatic and hand sanding, and plasma treatment
- Monitoring of post-surface treatment surface energy levels
- Quantification of surface preparation to ensure consistency and repeatability
- Troubleshooting bonding process inconsistencies and failures
Aircraft Application Example
A top aircraft manufacturer specializing in major defense applications needed to verify clean surfaces during their assembly process. Their surface preparation process included three steps: solvent wipe, hand abrasion, and then an additional, different solvent wipe. After these steps, an engineer measured the composite panel surface using a contact angle measurement - a simple, rapid, and quantitative test of surface energy. The results indicated a very high contact angle at 70°. In a typical preparation process situation, the higher the contact angle, the less reactive or more "dirty" the surface is. The lower the contact angle, the more reactive or "clean" the surface is. These measurements indicate surface preparedness prior to bonding.
Needless to say, this measurement tilted a few heads. Their surface showed a more contaminated surface after the specified preparation process than before.
By breaking down the process and conducting further analysis with additional contact angle measurements, the reason behind this occurrence began to become clear. It was only through meticulous examination that the answer to this mystery was revealed.
The original contact angle taken prior to any surface treatment read 70°. After the first solvent wipe, the contact angle read 50°. After the hand abrasion, 30°. So far, so good. But, after the final step of the solvent wipe, the contact angle hit the roof, reading higher than the original angle of 70°.
This led them to investigate the solvent itself, which was stored in an opaque container. The team poured the solvent into a clear beaker, surprised to find the cleaner was a charcoal color rather than clear. It was then that the team explained that they first used the solvent to soak other parts. Secondly, they poured it into a container to be reused in the surface bonding preparation process.
It was revealed that the manufacturer was using an extremely contaminated solvent to "clean" their surfaces. While the coloring of this solvent is obvious contamination, a solvent can be quickly contaminated and may lose its integrity well before showing physical proof. But, with accurate verification procedures, the quality of a solvent would never need to be left for guesswork.
Revolutionize Your Manufacturing with Surface Quality Inspection Technology.
Consumer Goods Must Meet the High Demands of an Informed Populace
The world of consumer goods is highly diverse, so consequently, manufacturing processes are even more varied. From golf clubs to paints to windows to consumer electronics, consumer goods products face a variety of stresses in the field that can include moisture, impact, contaminants, and environmental stresses. Manufacturers must produce a product to withstand those stresses.
Here’s a list of consumer product examples that require process controls to reduce the cost of manufacturing and prevent failures:
- Monitor carbon fiber bicycle frame flame and plasma treatment
- Monitor plasma treatments used in golf ball manufacturing
- Verify hydrophobic coatings, like fluoropolymers, in appliance manufacturing
- Monitor grit-blasting of metal surfaces such as golf clubs
- Evaluate metal cleaning prior to painting or bonding
- Verify hydrophilic coatings like resin laminates on surfboards
- Predict absorbency of skin care products on skins
- Validate conformal coatings on consumer electronic circuit boards like cell phones
- Verify bonding of composite bicycles
- Validate the surface of polymer window castings for bonding (these castings require perfect adhesion to maintain thermal efficiency and longevity)
- Validate flame treatment of appliances prior to painting or coating
- Verify the bonding strength of adhesives and tapes
- Guarantee uniform coatings of the linings in products like paint cans
- Guarantee quality control of laser welding processes in appliance manufacturing
- Guarantee strong bonds on solar panels
- Evaluate performance during the development of paints
- Guarantee printing ability on consumer goods packaging
Golf Club Manufacturer Application Example
Manufacturers often implement surface preparation methods to get more out of their bonding processes, but a new piece of surface treatment equipment is not the silver bullet these manufacturers are hoping for. Without a monitoring process that accurately verifies the molecular changes occurring on the surface of treated materials, if the variance in quality starts occurring, it is impossible to know what parameters need to be altered.
A high-end golf club manufacturer was developing a metal golf club head that needed to bond to a composite cover. To hold this bond, the manufacturer pre-treated the metal with a grit blaster. However, one day the surface cleanliness levels did not change as expected after treatment.
Brighton Science came in and took contact angle measurements on the metal parts in question. The contact angles were higher than expected, meaning the surface was not being cleaned properly.
Investigators from Brighton Science evaluated a grit-blaster used in a different area of the plant and then treated the metal using this other equipment. Much to the manufacturer’s surprise, the contact angle reduced dramatically before and after treatment, indicating the surface had been cleaned properly.
Get hands-on with your surface cleanliness with the Surface Analyst 5001.
The first grit-blaster turned out to have worn, or “dirty,” media being used to treat the metal. Consequently, the process was unable to properly create a surface that was prepared for bonding and even deposited further contaminants on the surface. Without a quantitative inspection of this process, the issue would have gone unnoticed until failure began. This quality assurance method was used not only to monitor the cleanliness of their media in the grit-blaster but also as a way to predict the bond strength of the golf club to fully ensure that each unit met or surpassed every performance requirement.
For more information on what steps to take to actually get to the root cause of adhesion and cleanliness issues, read our guide to reexamining your process holistically. The “Checklist: Adhesion Failure Root-Cause Analysis for Manufacturers” eBook is a step-by-step handbook for eradicating adhesion failure from the ground up.