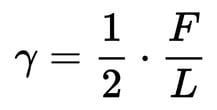Using adhesives in manufacturing is becoming increasingly common for building everything from massive machines to everyday tech devices. But companies' reliance on the science of adhesion to make sure their products work perfectly and look marvelous didn’t start there. In the centuries since the first ink was put to paper and the first paint was applied to metal, we have learned that we need to understand some basic laws of chemistry in order to ensure a liquid substance would reliably adhere to the surface of a solid substance. Among other things, these laws govern the properties we refer to as surface tension and surface energy.
This simple truth is at the heart of why surface tension and surface energy are so vital to manufacturing consistent, high-performance products efficiently and with low to no risk of failure.
All matter is made of molecules, and all molecules have varying degrees of attraction to other molecules. This intermolecular attraction is why the molecules of your drink, or even the ice in your drink, stay together in a glass or as a puddle on the tabletop rather than drift apart into the air. The attractive force of the molecules present at the surface of a liquid towards each other is called the surface tension of that liquid. Surface energy is the equivalent attractive force present between the molecules at the surface of a solid substance. This attractive force is what holds a substance together into a cohesive form. We usually use surface tension when referring to liquids and surface energy when referring to solids, but in reality, they are the same thing.
Surface tension and surface energy are important because they are measurable numbers that tell us how strong the attraction between molecules is. High surface energy (or tension) substances have high boiling points: they have to be heated up a lot in order to overcome the intermolecular attractions to form vapor or steam.
Rethink your adhesion manufacturing processes with Surface Intelligence.
Let’s go back to ink on paper as an example of surface tension in play. We want ink to spread and stick to a surface when applied through a printer or even our fancy feather quill. If the molecules in the ink are attracted to each other strongly (exhibiting high surface tension), when they come in contact with the surface, they won't want to spread out and stick to the surface of the paper or plastic bottle, or another material we want to emblazon with our company’s logo. They will prefer to stay associated with each other due to the strong intermolecular attraction. On the other hand, the molecules in a low surface tension liquid are not attracted strongly to each other; they will instead tend to spread out and stick to the surface you want to print to.
The liquid will tend to spread and stick because the attractive forces from the molecules at the surface of the paper or plastic are pulling on the molecules at the surface of the ink. This interface is where surface tension and surface energy combine to result in adhesion.
This vital information about how liquids react to solid surfaces is one of the reasons surface tension and surface energy have very real implications in manufacturing processes. Understanding this relationship and the chemical processes taking place at the interface of surfaces can have a major impact on process optimization and process control.
What is Surface Tension?
As we stated above, surface tension is the attractive force of the molecules present at the surface of a liquid towards each other. But to understand it a little more deeply, it’s helpful to think about what surface tension isn’t.
For more information about controlling variables that often go overlooked yet still affect the outcomes of your manufacturing process, download our free eBook: Predictable Adhesion in Manufacturing Through Process Verification
The forces we call surface tension are not like gravitational forces. The key difference is the gravitational forces scale with mass, and therefore the gravitational force between two molecules is miniscule to negligible. The reason we all stick to the Earth so well is that the planet has an enormous mass creating a very strong gravitational force on us.
Surface tension, on the other hand, is due to the attraction of, unlike, electrical charges that result from the distribution of electrons around the molecules. Surface tension is literally the force required to overcome these attractions and pull apart molecules that are present on the surface of a liquid. What that means is, if you were to draw an imaginary line on the surface of a liquid film or bubble and then attempt to pull it apart, the force required to pull and split the liquid at that line, divided by the length of the line, would be the surface tension of that liquid.
Surface Energy Formula
We can express surface tension as a simple mathematical formula:
Where the surface tension (γ) is the force across an imaginary line divided by 2 times the length of the line. The 2 is necessary because when we make a split in the surface of a bubble, we are actually pulling apart the molecules on two surfaces: the inner surface and the outer surface.
How Can Surface Tension be Measured
It's hard to imagine being able to pull apart the surface of a liquid bubble. But there are a couple of ways to measure this force fairly easily. One of them is to measure the force required to pull a thin wire ring out of a container of liquid. Do this very slowly (so liquid viscosity doesn't affect the results), and the force required to just detach the ring from the surface divided by the circumference of the ring (divided by 2 because there is an inner and outer circumference) gives the surface tension. (This is the basis of the du Nouy Ring method; there is a correction factor needed to account for the liquid meniscus shape, but it's all been worked out). Another measurement is based on the pressure necessary to create and detach a bubble from the end of a needle immersed in the liquid (Maximum Bubble Pressure method); here, we are measuring the force necessary to create a liquid surface. More information on these techniques is readily available.
Surface tension is not the same as viscosity, although sometimes people confuse the two. Viscosity is a measure of the resistance to flow or, more precisely, how hard it is to move liquid molecules past each other. It's related to the friction between molecules. Viscosity is why it takes muscle to push your hand through water; it's the friction you feel as the molecules slide past each other. It’s fairly easy to measure viscosity. One common measure of viscosity is the time it takes a ball bearing of a certain size to travel a fixed distance when dropped into a liquid. Again, surface tension and viscosity reflect very different properties. Water has high surface tension but low viscosity. Mineral oil (like baby oil) has high viscosity but low surface tension.
Factors Affecting Surface Tension
Anything that affects the attraction between molecules affects surface tension. For example, water molecules attract each other strongly because they are highly polar molecules: the plus and minus charges on the hydrogen and oxygen atoms attract the opposite charges on neighboring molecules, and it takes a lot of force to pull them apart (this is why water has a very high boiling temperature even though it is a small molecule). There are other types of intermolecular attractions besides those that exist between polar molecules.
We’ll get more into those in another article but suffice to say the attractions that exist between the polar parts of molecules are generally quite strong, and perhaps more importantly they can be manipulated and changed on solids like polymers by surface treatments. This is why we frequently treat packaging materials with a corona or flame to improve ink adhesion.
For manufacturing purposes, we are frequently trying to understand the properties of materials we can control to make them come together and stick together reliably - in a way that is measurable. Knowing the surface energy lets us predict how substances will interact with each other. It gives us a window into chemical processes we can control. Measuring these processes lets us set standards; controlling these standards adds value to our manufacturing processes by allowing us to predict outcomes.
Surface tension changes depending on the composition of a liquid. For an illustration from everyday life (for some of us, anyway!), let’s go to a popular class of mixtures of liquids with different surface tensions, ethanol + water. Water has a relatively high surface tension, much higher than alcohol (because water molecules are more polar). When the two are combined into a beverage like wine, the resulting liquid has a surface tension somewhere between the two, lower than water but higher than ethanol.
Another property of ethanol is its high vapor pressure compared to water: an ethanol/water mixture will decrease in alcohol content as it sits in a glass. As the alcohol content decreases, the surface tension increases.
 When a glass of wine is swirled around, the thin film left on the sides of the glass quickly loses ethanol to evaporation. This increases the surface tension. Because this film has higher surface tension, it pulls on the wine that's down in the glass, and the result is the 'fingers' or 'tears' of wine that we see climbing the sides of the glass! Wines with higher alcohol content show this effect more strongly because it loses more alcohol to evaporation and increases surface tension even more.
When a glass of wine is swirled around, the thin film left on the sides of the glass quickly loses ethanol to evaporation. This increases the surface tension. Because this film has higher surface tension, it pulls on the wine that's down in the glass, and the result is the 'fingers' or 'tears' of wine that we see climbing the sides of the glass! Wines with higher alcohol content show this effect more strongly because it loses more alcohol to evaporation and increases surface tension even more.
Wine fingers are an example of what is called the Marangoni effect, which is the phenomenon of the flow of liquid caused by surface tension gradients.
On a side note, the presence of tears in your alcoholic beverage has nothing to do with quality, only with the alcoholic content (or proof) of the beverage.
What is the Difference Between Surface Tension and Surface Energy?
As we stated in the introduction, these two terms really only differ as a matter of semantics. But, surface tension is typically used in reference to the attractive forces of liquid molecules while surface energy is typically used in reference to the same between molecules of a solid substance. Their relation to one another is useful in measuring both and then utilizing one as a powerful process control mechanism in manufacturing.
Both surface tension and surface energy are expressions of how strongly molecules on a surface are attracted to each other. But when we’re talking about their role in creating predictably strong adhesive bonds, they each have a distinct function.
Measuring surface tension can be as easy as measuring the pressure required to blow, as we described earlier. However, to measure the surface energy of a solid, we need to use indirect methods. For example, we can measure the tendency of liquids to spread (wet out) or bead up on a surface and can infer what the surface energy is from the shape of the liquid drop. The angle created by the edge of a drop of liquid with the solid surface correlates with the surface energy of the material surface.
Manufacturers who use liquid products like paints and adhesives as a means to assemble and finish products ranging from microchips to military jets can use a knowledge of surface energy to control how well and how consistently those liquids will stick to the surfaces of their products. The adhesives and coatings manufacturers have done their part, but control of the surface energy is the other crucial component in the quest to successfully bond or coat a product.
The surface energy of materials that have adhesives, coatings, and inks applied to them needs to be known and controlled in order to have the assurance that the bond will be strong. If the surface energy is at the appropriate levels for the application, and the adhesive manufacturer controls the surface characteristics of their product, then manufacturers can rest assured their assembly will meet every performance test that relies on those bonds holding fast.
Brighton Science specializes in partnering with manufacturers who want to control their adhesion processes, and we frequently use surface energy measurement to help them achieve that control. We offer a whole line of instrumentation that makes it easy to measure and control the surface energy of materials directly on the production line. Our technology allows manufacturers to quantitatively evaluate the quality of their bond surfaces through a rapid test that definitively indicates if their surface is ready for bonding or coating.
For more information about controlling variables that often go overlooked yet still affect the outcomes of your manufacturing process, download the eBook: “Predictable Adhesion in Manufacturing Through Process Verification.”