This is part two of a two-part series explaining Brighton Science's approach to helping companies build reliability into their cleaning and adhesion processes through consultation and implementation of novel inspection equipment. These two articles are based on this technical paper written by Dr. Giles Dillingham about the best way to measure surface quality.
For a brief recap of the first part of this deep dive into the science behind the best and most efficient methods of process control in manufacturing, let's go over a couple of crucial points to remember about the fundamentals of adhesion:
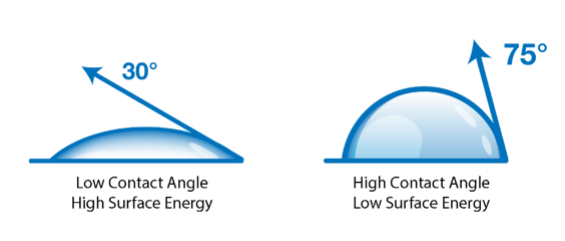
Materials (e.g., metals, polymers, wood, etc.), substances (adhesives, coatings, inks, paint, etc.), and the objects they form when assembled stick to each other for a simple reason: the molecules they are made of attract each other. Moreover, in most situations, this attraction can be most easily quantified and accurately measured through contact angle measurements.
These measurements offer crucial information to manufacturers about the state of chemical cleanliness or level of treatment of their surfaces, which is critical to control for any process involving bonding, coating, sealing, printing, painting, soldering, sintering, and a host of other methods reliant upon interfacial adhesion.
That's where the first article left off, but to be certain you have the most effective surface quality verification system, it's helpful to go deeper into the science.
Rethink your adhesion manufacturing processes with Surface Intelligence.
Say you're a manufacturing company that produces electronic components for aerospace applications like satellite tracking devices or navigational equipment. Or perhaps you build medical devices that involve coatings on glass diagnostic tools. You might even be a sports equipment manufacturer who wants to ensure that your new logo printing process works every time.
After reading part one of this series, you decide to invest in a contact angle measurement system to track surface quality throughout your production process. How do you know which one to go with? No matter how you slice it, contact angle is a contact angle, right? Well, yes and no. Some approaches demand multiple fluids, while others use only one.
For more information on how Brighton Science partners with companies who want to make strategic decisions using data from smart tools, download our eBook outlining our approach. Get the Guide: Guide to Adhesion Science for Flawless Manufacturing
To parse out the differences in how various fluids used in contact angle measurement systems interact with surfaces along with the type of information that each provides, we need to dig a little further into what molecular attraction looks like at an atomic level and how that changes throughout surface treatment processes.
Why Substances Attract Each Other
The attraction between all materials and substances is ultimately based on the attraction between the negative and positive charges of the electrons and protons that make up the atoms of the molecules in all materials and substances. There are two ways that the negative and positive charges of electrons and protons affect how molecules bond. One of these ways is based on random, ever-shifting movements by electrons encircling the nuclei of the atoms that make up the molecule.
To illustrate this, think about holding a molecule in your hand and looking at it like you would any small object. You would see a cloud of negative charge (the electrons) defining the shape on the outside, with the nuclei (containing the positive protons) hidden inside this electron cloud. The electron cloud is always in a random, fluid motion. While it has a well-defined average shape, because of the random movement of the electrons, it can be concentrated over one part of the molecule for one instant and concentrated over another part in the next instant.
When the electrons are momentarily concentrated over one part of the molecule or another, the abundance of electrons will give that part of the molecule a predominantly negative charge, leaving the rest of the molecule with a slight (and compensating) positive charge. This charge distribution is called a temporary dipole, which exists in all atoms and molecules.
Because positive and negative charges attract each other, the temporary dipoles of atoms and molecules attract each other. The attraction is weak, but it exists between all molecules. The attraction between temporary dipoles is why, for example, vegetable oil doesn't evaporate but exists as a liquid in an open container. These forces are called dispersive forces because they result from fleeting dispersions of the electron cloud around a molecule.
Although present between all substances, these dispersive attractions between temporary dipoles are very weak. Permanent dipoles can also exist if the molecule has the proper structure, and these can give rise to much greater attractive forces between molecules. Take, for instance, a single molecule of vitamin C: C6H8O6, which has six carbon atoms, eight hydrogen atoms, and six oxygen at ms. Oxygen atoms have a higher number of protons (which are positively charged) than carbon or hydrogen. Therefore, the parts of the molecule with oxygen atoms will tend to attract more of the electron cloud (which is negatively charged) than the parts of the molecule where the carbon and hydrogen atoms reside, resulting in a part of the molecule that is always charged more negatively than the other parts: such a molecule has a permanent dipole.
The polar forces of attraction between permanent dipoles can easily be ten times stronger than the weaker attraction between temporary dipoles. This is why a substance like ethanol (with a permanent dipole) is a liquid at room temperature, while a substance with similar mass but only temporary dipoles (e.g., propane) is a gas.
The strong attractions between permanent dipoles are essential for adhesion. Polymers like polyethylene and polypropylene have no permanent dipoles and can only attract substances through weak temporary dipoles: inks and adhesives don't stick well to them unless we treat the surface with flame, corona, or plasma to introduce structures with permanent dipoles. Other polymers, such as polyesters, polycarbonate, or acrylics, have these permanent dipoles as part of their structure. Adhesion to these polymers can often be achieved with no treatment beyond careful cleaning.
Total Surface Energy and Its Components: Dispersive and Polar
You might have heard the term total surface energy come up in conversations about controlling bonding, coating, or other adhesion processes. Total surface energy refers to the totality of attractive forces that are available to act between the surfaces of materials and substances. Knowing the total surface energy of your material can be helpful for getting a complete picture of the forces at play on your surface. However, measuring total surface energy for the purpose of controlling surface treatment and cleaning processes can be inefficient or even misleading. It can be much better to hone in on the component of surface energy that is important to adhesion and characteristic of the changes occurring during surface preparation procedures.
We mentioned above that polymers possess permanent dipoles, which is why they can frequently adhere well to adhesives, paints, and inks with no additional treatment. For other polymers, like polypropylene or polyethylene, oxygen or nitrogen can be grafted to the polymer surface by a plasma, corona, or flame treatment, providing significantly improved adhesion because the polar attractive forces have been increased by the addition of elements that will pull electrons to one area of the molecule.
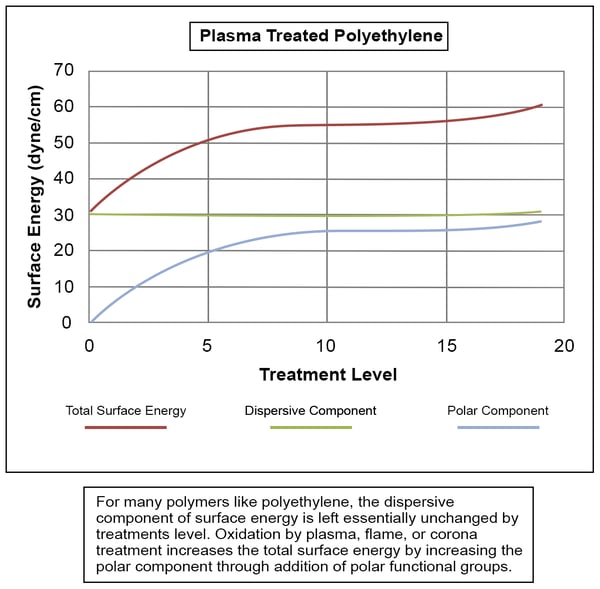
So, when we talk about measuring surface energy as an approach to process control in manufacturing, a couple of things begin to become apparent about the differences in constituent forces within surface energy. As an example of these differences, the chart below shows how the polar and dispersive components of the surface energy of polyethylene change as the total surface energy is increased by plasma treatment. It's evident in this case that the polar component is affected most by plasma treatment; the dispersive component is affected very little (if at all). This is frequently the case.
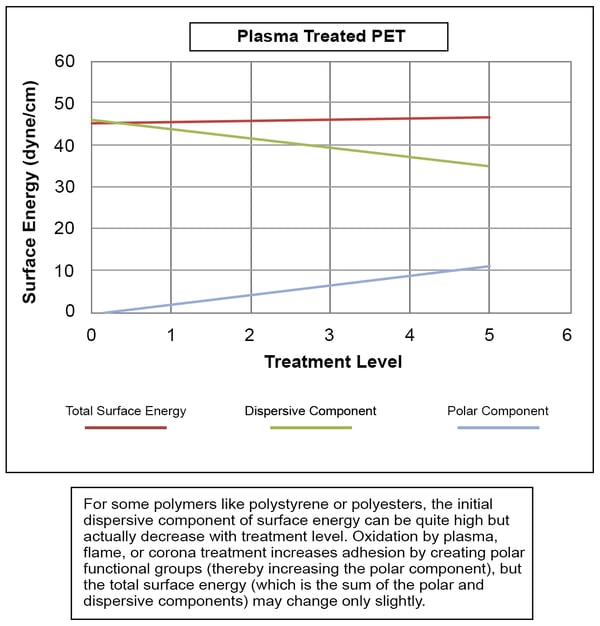
The following chart illustrates the dangers of looking at total surface energy without breaking it into its constituent forces. The total surface energy of the polyester that was plasma treated to get these results remains relatively constant in this instance. The increase in the polar component is negated mainly by a decrease in the dispersive component. These surfaces still show excellent improvement in adhesion because the polar interactions with the coating or adhesive override the dispersive interactions. In this case, if you were measuring the total surface energy as opposed to the polar component of surface energy, you might assume that the material had not been adequately cleaned or treated even though the forces necessary to create strong, sustainable bonds had increased sufficiently. This fact would be hidden by the relative lack of change in total surface energy, and this could lead to unnecessary additional steps or even overtreatment, which could damage the part being processed.
Measuring Surface Treatment via Contact Angle Measurements
To determine the contribution of each of the two different forces to the total surface energy of a substance, we must take contact angle measurements with at least two different liquids that have known (and different) polar and dispersive components to their own surface tension (see the part 1 of this article for information about liquid surface tension and how it relates to surface energy).
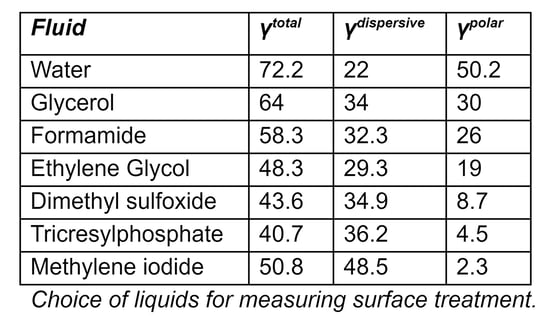
The accompanying table above shows liquids that are commonly used for measuring surface energies, along with the dispersive and polar components of their surface tensions. The two most common liquids used for measuring total surface energy are water (a highly polar liquid) and diiodomethane (methylene iodide in the table, which is a highly dispersive liquid with very little polarity).
Using two liquids is the basis for most commercial methods of measuring total surface energy using contact angle techniques. However, the accuracy of surface energies measured this way is limited by the uncertainty in how well we know the polar and dispersive components of the liquid surface tension and by how accurately we can measure the contact angles. The precision of the surface energies calculated from contact angle measurements is greatly improved if more than two fluids are used. It's important to note that although many commercial software packages use contact angles measured from two fluids to calculate surface energies, the error between the true total surface energy and what these liquids measure can be quite large. Essentially, this fact means that companies who want to help manufacturers measure total surface energy using contact angle should actually use contact angle measurements of several more liquids to get a more accurate measurement.
The most important thing to remember here is that even if you used half a dozen liquids and got a very accurate measurement of total surface energy, this data may not be that useful in building better, more reliable products.
 Suppose you are attempting to measure the aspect of surface energy that gives you the most reliable information about how reactive and ready for bonding your surface is, the aspect that has changed the most during treatment. In that case, you need to understand this principle: the polar part of a liquid only interacts with the polar part of a surface, and the dispersive part of a liquid only interacts with the dispersive part of a surface. Polar liquids (like water, as shown in the table above) have contact angles that respond strongly to the polar component of surface energy. When the polar forces go up, the contact angle of water goes down as the water is highly attracted to the surface and wets out. Similarly, if the polar forces decrease, the water is more attracted to itself, producing a high contact angle.
Suppose you are attempting to measure the aspect of surface energy that gives you the most reliable information about how reactive and ready for bonding your surface is, the aspect that has changed the most during treatment. In that case, you need to understand this principle: the polar part of a liquid only interacts with the polar part of a surface, and the dispersive part of a liquid only interacts with the dispersive part of a surface. Polar liquids (like water, as shown in the table above) have contact angles that respond strongly to the polar component of surface energy. When the polar forces go up, the contact angle of water goes down as the water is highly attracted to the surface and wets out. Similarly, if the polar forces decrease, the water is more attracted to itself, producing a high contact angle.
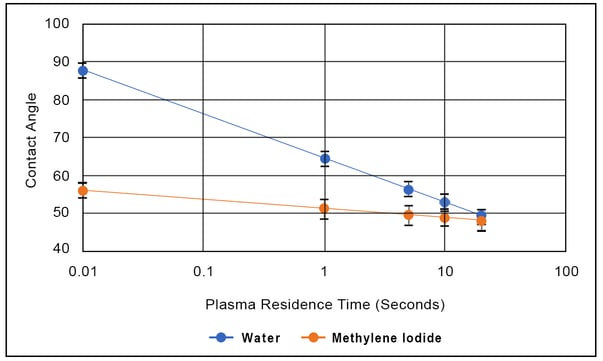
Conversely, liquids whose surface tension is mostly dispersive (methylene iodide, for example) have contact angles that do not change very much as the polar component of the surface energy changes. The accompanying figure shows the effect of plasma exposure time on the contact angles for water and methylene iodide on polypropylene. The contact angle for methylene iodide changes a small amount with plasma treatment. This is a poor metric for process control since the contact angle numbers hardly change as the strongest forces of attraction are significantly modified. Furthermore, the toxicity of methylene iodide makes it a poor choice for deploying in a manufacturing environment.
On the other hand, this Figure shows that the contact angle of water is very sensitive to treatment level: the treatment is primarily changing the polar character of the surface, and water is a highly polar liquid. There is a one-to-one correspondence between water contact angle, contamination level, and surface treatment level that makes it, in many ways, an ideal metric for controlling surface treatment. When you can see precisely what is changing during treatment, you can optimize that process to get the desired results.
Revolutionize Your Manufacturing with Surface Quality Inspection Technology.
In summation of both articles: all substances attract all other substances through at least two basic mechanisms: the weak dispersive forces that exist between all atoms and molecules regardless of structure and the strong polar forces that depend on the details of the molecular composition and structure. In most cases, the dispersive component of intermolecular interactions contributes little to adhesion and changes little with treatment. Because some materials actually show a decrease in the dispersive component of surface energy with treatment, using the total surface energy as a metric to control surface treatments can be misleading.
The polar component of intermolecular attractions is responsible for the adhesion enhancement of surface treatments such as corona, flame, and plasma treatments and can be controlled with many nuances. A simple water-only contact angle measurement frequently fills all the requirements for a sensitive process control metric because it can give valuable information about the polar forces of attraction.
For more information on how Brighton Science partners with companies who want to make strategic decisions using data from intelligent tools that offer insight that directly correlates to the most important changes to surfaces during a cleaning and treatment process, download our eBook outlining our approach. You can get the guide: Guide to Adhesion Science for Flawless Manufacturing.